FDA Compliance at Kamerycah, Inc.
FDA Compliance at Kamerycah, Inc.
At Kamerycah, Inc., we operate our quality control systems in alignment with U.S. FDA regulations (21 CFR), ensuring safety, consistency, and transparency across all health supplement products.
Raw Material Control
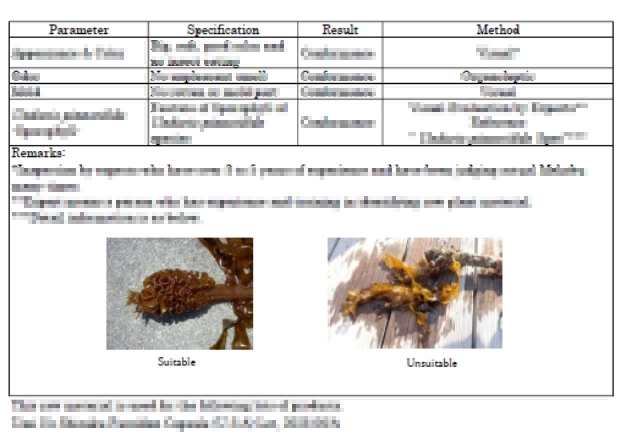
All raw materials are verified based on predefined specifications. Identity, purity, and safety are confirmed through rigorous analytical testing before any material enters the production process.
Certificates of Analysis (COA) are reviewed for every lot to ensure consistent quality.
Testing performed on all raw materials:
- Heavy metals testing (ICP-MS)
- Microbiological testing
- Fucoidan (fucose) content verification


In-Process Quality Control
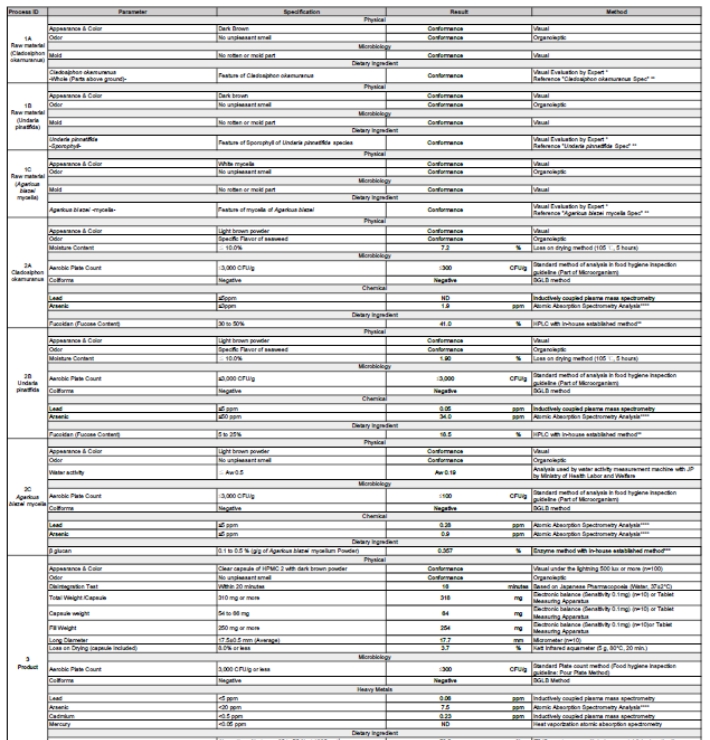
Intermediate materials are monitored throughout the production process. Critical parameters are controlled at every stage to ensure batch-to-batch consistency.
All manufacturing is conducted under GMP-certified facilities, operated by trusted partners with verified compliance records.
GMP-Certified Manufacturing Facilities:
- Biholon Co., Ltd. (USA cGMP / Japan GMP)
- Yonedakeiteisha Co., Ltd. (Japan GMP)




Finished Product Quality Assurance
Each batch is tested against predefined specifications before release. Final products are evaluated for safety, consistency, and composition to confirm they meet all required standards.
Testing performed on finished products:
- Heavy metals testing
- Microbial safety verification
- Content uniformity


Proprietary Analytical Testing
In addition to standard testing methods, Kamerycah utilizes advanced analytical techniques to ensure the highest level of product integrity across every production lot.
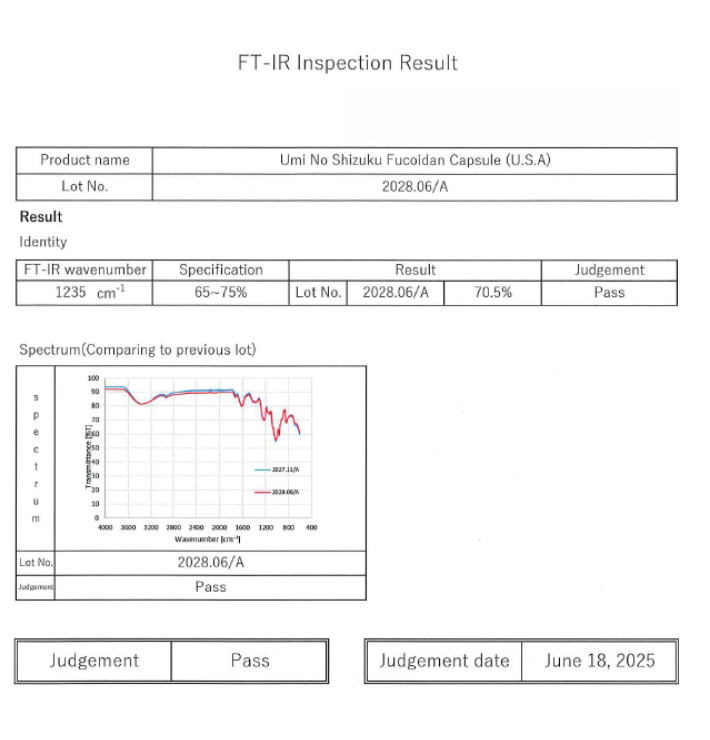
FT-IR (Fourier Transform Infrared Spectroscopy)
- Used to verify structural consistency of fucoidan
- Enables comparison across production lots
- Supports identity and quality verification beyond standard assays
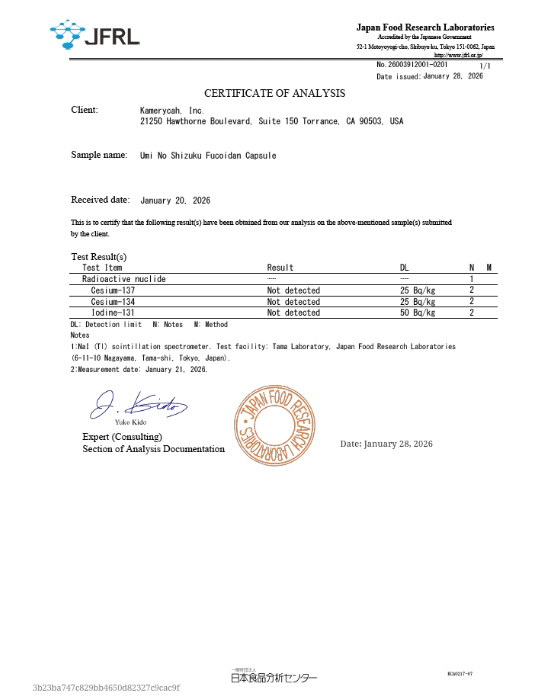
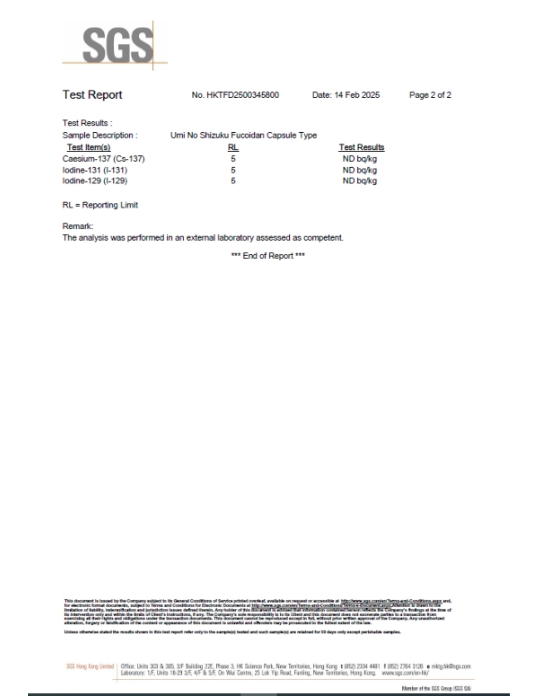
Third-Party Safety Verification
Kamerycah products are tested by independent third-party laboratories, including SGS and the Japan Food Research Laboratories (JFRL), to verify product safety.
These tests confirm that levels of radioactive substances, including cesium and iodine, are below established safety standards. Certificates are issued to validate compliance and ensure product safety.
JHFA Certification
The JHFA Mark is a certification issued by the Japan Health and Nutrition Food Association (JHNFA), a public interest incorporated foundation in Japan.
- Awarded only to health food products that meet strict safety and quality standards
- Products that do not meet these criteria are not certified
- Provides independent third-party verification of product quality
- Fucoidan Umi No Shizuku has been certified under these standards
This certification ensures that our products meet recognized standards for safety and quality, providing confidence and peace of mind.


Packaging Material Quality Control
All packaging materials are verified against predefined specifications before use. Materials such as capsules, containers, and labeling components are evaluated to ensure suitability, safety, and consistency.
Packaging materials are inspected for appearance, integrity, and conformity prior to production. Compatibility between packaging materials and product contents is confirmed to maintain product stability.
All packaging materials and processes are managed to protect product quality and ensure consistency from manufacturing through final delivery.

Import Compliance & Responsibility
Kamerycah operates under a Foreign Supplier Verification Program (FSVP). Supplier qualification and documentation are strictly controlled, and compliance is managed by certified personnel.
Controlled storage environments are maintained to preserve product stability and quality from import through final distribution.


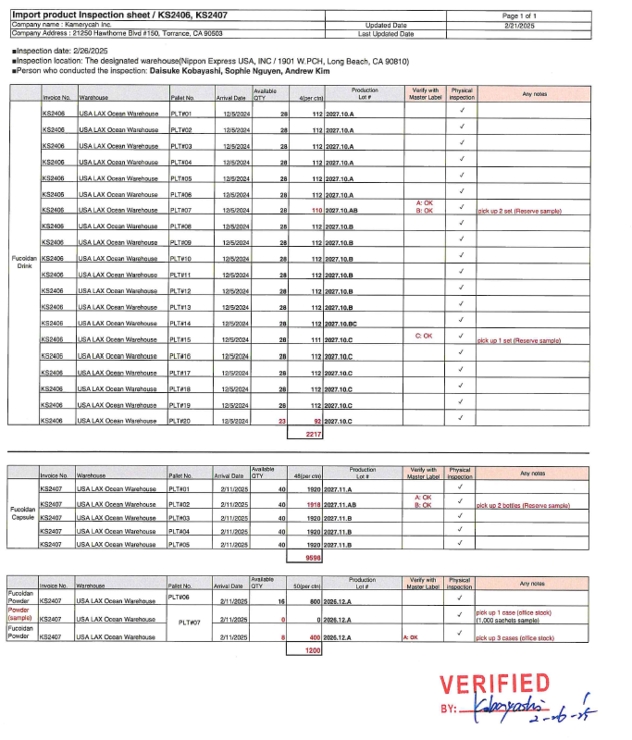
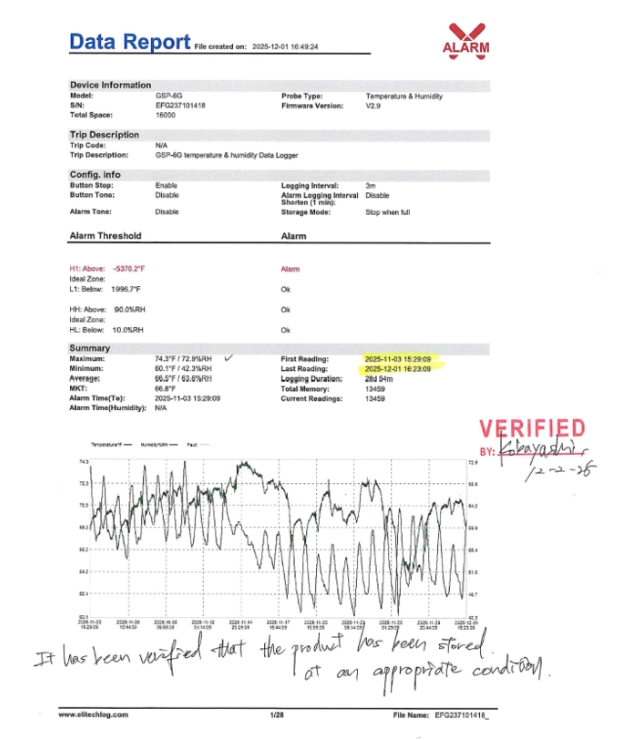


At Kamerycah, Inc., quality is not defined by a single number or claim. It is built through a structured system that ensures safety, consistency, and regulatory compliance at every stage — from raw materials to finished products.
Kamerycah, Inc. is committed to delivering products you can trust with confidence.

